 
Each forward-looking statement is subject to risks and uncertainties that could cause actual results to differ materially from those expressed or implied in such statement. In some cases, you can identify forward-looking statements by terms such as "expect," "continue," "plan," "intend," "will," or "typically," or the negative of these terms or other similar expressions, although not all forward-looking statements contain these words, and the use of future dates. This press release contains forward-looking statements under the Private Securities Litigation Reform Act of 1995, including statements about the benefits of Vapotherm’s products. Legal Notice Regarding Forward-Looking Statements 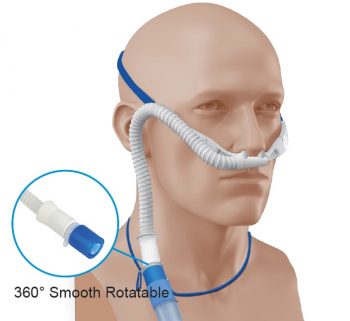
The information contained on, or that may be accessed through, Vapotherm’s website is not incorporated by reference into, and is not a part of, this document. Accordingly, investors should monitor the Investor Relations section of Vapotherm’s website, in addition to following Vapotherm’s press releases, Securities and Exchange Commission ("SEC") filings, public conference calls, presentations and webcasts. Vapotherm intends to use this website as a means of disclosing material, non-public information and for complying with Vapotherm’s disclosure obligations under Regulation FD. Vapotherm routinely posts important information for investors on the Investor Relations section of its website. While being treated, patients can talk, eat, drink and take oral medication. The HVT 2.0 and Precision Flow systems’ mask-free interface delivers optimally conditioned breathing gases, making it comfortable for patients and reducing the risks and care complexities associated with mask therapies. It allows for the fast, safe treatment of undifferentiated respiratory distress with one tool. For more information, visit Vapotherm high velocity therapy is mask-free non-invasive respiratory support and is a front-line tool for relieving respiratory distress-including hypercapnia, hypoxemia, and dyspnea. Over 4.0 million patients have been treated with the use of Vapotherm high velocity therapy® systems. The Company develops innovative, comfortable, non-invasive technologies for respiratory support of patients with chronic or acute breathing disorders. (NYSE: VAPO) is a publicly traded developer and manufacturer of advanced respiratory technology based in Exeter, New Hampshire, USA. Jessica Whittle, Chief Medical Officer at Vapotherm said, "research like this helps clinicians stop and rethink their understanding of ventilation and to consider using high velocity therapy as a first line treatment for patients needing more than traditional oxygen." "This reaffirms that not all high flow devices are the same and shows the value of high velocity therapy for clearance of CO 2 and flush of the upper airway," said Brian Lawrence, Chief Technology Officer at Vapotherm.ĭr. It is also important for patients who may not breathe with optimal efficiency such as those who are sedated for some medical procedures. This finding correlates with the known clinical benefits of Vapotherm’s High Velocity Nasal Insufflation (HVNI) therapy and is especially significant for patients with chronic obstructive pulmonary disease (COPD) and asthma. The modeling confirmed that the higher velocity generated by the Vapotherm design results in improved CO 2 flush compared to other cannulas. Vapotherm’s unique small bore design generates higher gas velocity distinguishing it from other forms of high flow cannulas. This work validates findings presented at the 8 th Thermal Fluids Engineering Conference in March 2023 in the presentation titled "Cannula Oxygen Therapy." Researchers utilized computational fluid dynamics to evaluate and describe the impact of cannula design on air flow and gas exchange in the upper airway. Computational fluid dynamics uses advanced computer algorithms to understand gas flow characteristics in changing and complex systems. The full article will be published in the December issue of J. (NYSE: VAPO), a global medical technology company focused on the development and commercialization of its proprietary Vapotherm high velocity therapy® products, which are used to treat patients of all ages suffering from respiratory distress, today announced the online publication of "Validation of Human Upper Computational Fluid Dynamics Model for Turbulent Mixing" in the Journal of Fluid Dynamics. EXETER, N.H., October 04, 2023-( BUSINESS WIRE)-Vapotherm, Inc. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
